
Mission
"In close cooperation with our clinical advisors and partners in industry we fight costs, illness and deaths caused by stent and catheter related emergencies and infection. We fight the progression of bacterial resistances caused by the wide use of antibiotics, silver particles and other biocide substances. We improve patients' quality of life and at the same time help to lower healthcare related costs."
Situation

Infection, encrustation and blockage of indwelling catheters, ureteral stents and nephrostomy tubes are significant medical and health-economic problems. Because of these problems
- about 50% of patients with permanent urinary drainage devices present as emergency cases during their course of disease
- 13000 patients die every year
- up to 1.3 Billion Euro additional costs are produced per year in Western Europe and a similar amount in North America.
Source: Riedl CR, International Journal of Antimicrobial Agents; Klevens RM, Public Health Reports
Challenge
Patients with indwelling urinary devices are often treated with antibiotic medication. However, the use of antibiotics has not proven to suppress encrustation and their wide use harbours the risk of promoting bacterial resistances.
To use coated stents and catheters is another approach. But also antibiotic, silver and other biocide coatings lead to resistances. Furthermore these coatings accumulate cell material of dead bacteria on the surface, and that makes them ineffective after a short period of time. The same is true for drug eluting coatings, as their drug reservoir only lasts for one or two days. All these approaches could not proof to be effective in clinical use.
Breakthrough
UroNova's unique patented technology protects stents and catheters from biofilm formation and encrustation, resulting in fewer blockages of the urine drainage and injuries during removal. And this is accomplished without using antibiotics, silver particles or any other biocide substance. Proven, millionfold in clinical every-day practice.
No drug elution takes place, so no systemic side effects are caused. The coating's effectiveness is not limited in time, as it is long-term stable. Biocompatibility is unparalleled.
The CLEANUS Study Results
In a double-blind study of 100 patients, UroNova’s innovative coating, which uses a stable, negative surface charge to repel bacteria, produced significant improvements over conventional stents:
- Severe Encrustations: Dropped from 13% to 0%.
- Antibiotic Use: Reduced by 78%.
- Pain Management: 66% less pain reported, with a 49% reduction in pain medication.
- Recovery: 75% fewer days spent in bed and 90% less impairment of general well-being.
- Infection Markers: 86% fewer patients showed elevated leukocyte counts.

Left: Uncoated conventional stent with severe encrustation. Right: Coated CLEANUS stent.
Click on the video to learn about the process of biofilm formation and encrustation and how UroNova's coating is providing protection.
Services
UroNova is offering contract coating services for the medical device industry.
With over 20 years of experience in development, regulatory approval and production of coatings we can support you in all steps of the product lifecycle.
Patent protection of our core technologies is another key strength of UroNova and a fundamental advantage for all our customers.
Benefits
Medical devices refined with UroNova's unique coating technology offer various clinical, technological and financial benefits, for the distributor, healthcare provider, insurer and patient:
Clinical
- Proven millionfold in clinical practice
- No promotion of bacterial resistance
- No drug elution and associated side effects
Technological
- Available on most device materials
- Complete coverage in- and outside
- Validated quality control and testing
Health Economy
- Less catheter blockage and device exchanges
- Cost savings due to less emergencies
- Less complications and medication
Business
- Differentiation from competition
- Door opener to premium segment
- No costly development process
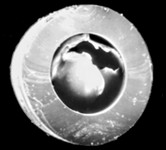
Left: Uncoated ureteral stent after 4 weeks indwelling time, massive biofilm formation.
Right: Ureteral stent with UroNova coating after 4 weeks indwelling time, in the same patient. No significant deposits.
From: Riedl et al., Int J Antimicrob Agents, 2002 (19)
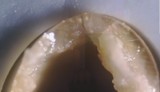

Left: Uncoated silicone nephrostomy balloon catheter after 2 weeks indwelling time. Complete blockage by encrustation.
Right: Silicone nephrostomy balloon catheter with UroNova coating after 6 weeks indwelling time, in the same patient. No significant deposits.
From: Tenke et al., Int J of Antimicrob Agents, 2004 (23)
Current clinical studies:
Awards (external content):




